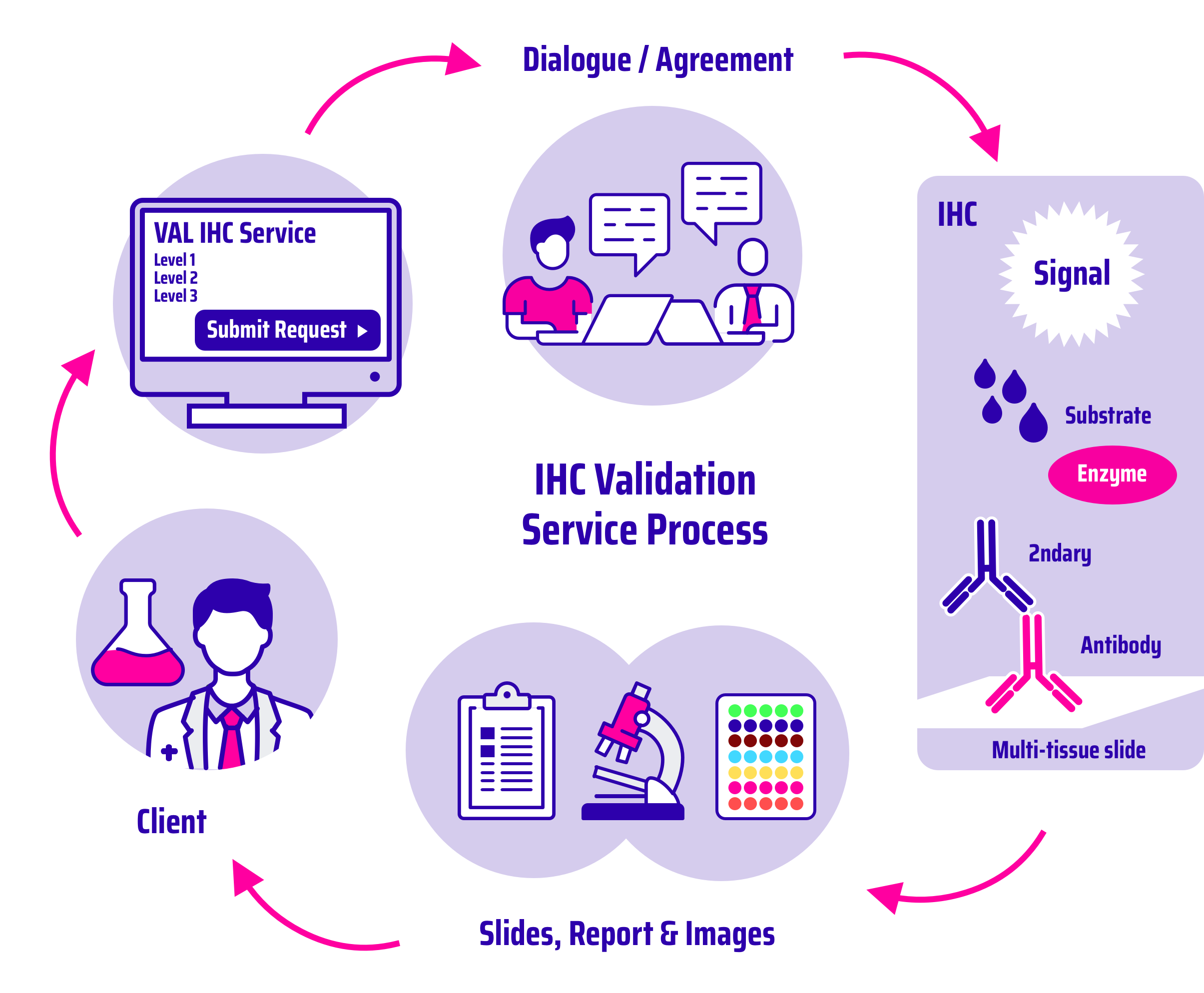
Immunohistochemistry (IHC) is a powerful technique which exploits the specific binding between an antibody and its antigen to detect specific targets in cells and tissue. IHC is widely used for the diagnosis of many diseases. We are committed to ensuring that your antibodies are appropriately validated to increase their research and clinical value.
Important note: all tissue specimens were received fresh in the diagnostic histopathology laboratory, reviewed and marked on the appropriate haematoxylin and eosin-stained slide by an expert pathologist.

Our IHC validation portfolio is tiered to cater to clients needs and requirements. As well as offering IHC antibody characterisation against multi-tissue microarray, we also provide characterisation for antibodies with unknown antigen retrieval conditions, unknown optimal dilution, or unknown expression profiles.
This service can complement one of our custom antibody production packages to provide a complete end-to-end solution. Alternatively, you can still supply your own antibody for assessment by our highly specialised team.
Our IHC platform’s key advantages:
- Standardised procedure – IHC is performed in an externally audited NQUAS (National Quality Assurance Standards) accredited laboratory.
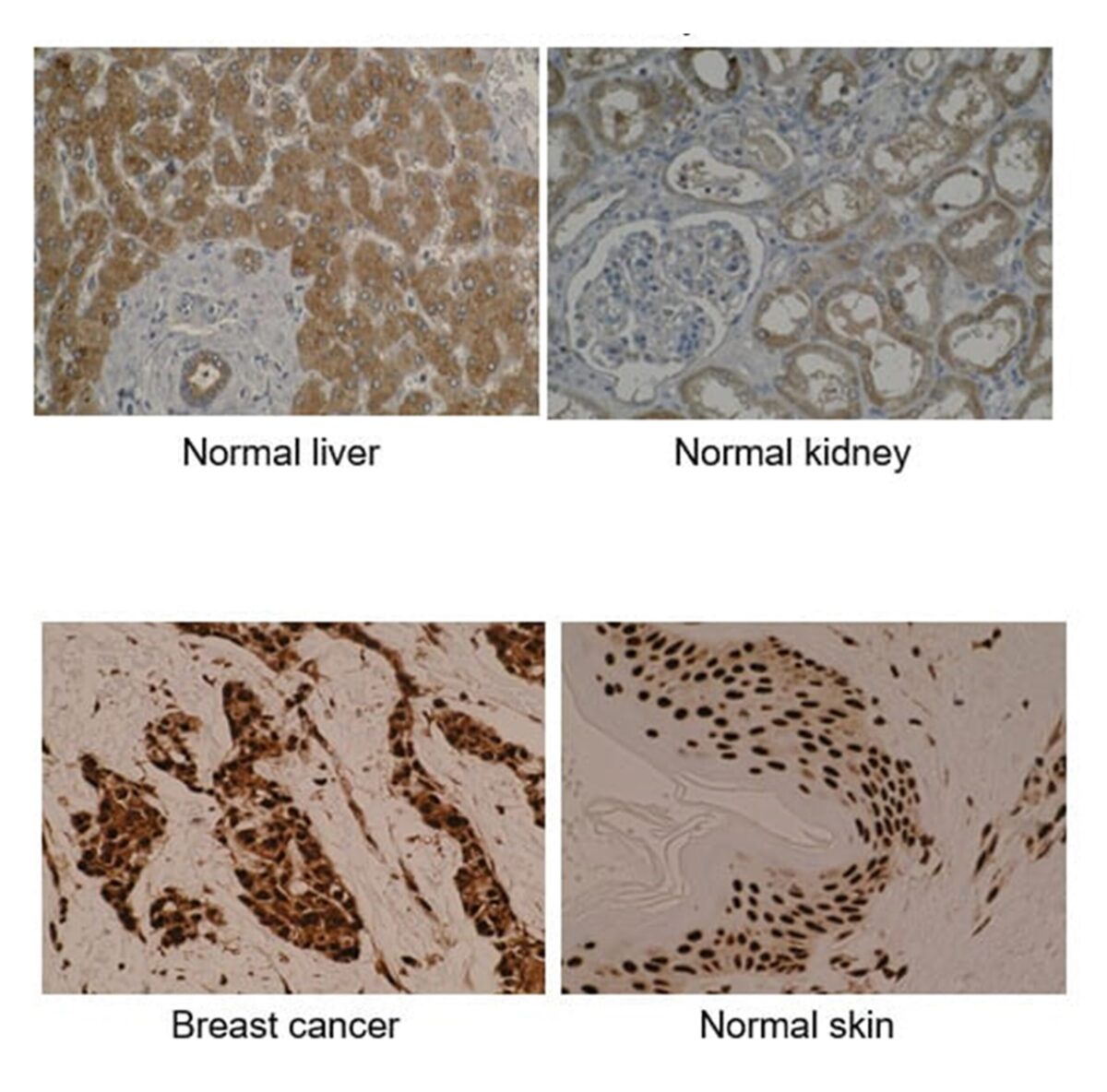
- Comprehensive – the multi-tissue microarray is comprised of human normal (n=53) and tumour (n=72) tissues from 20 different major organs.
- Access to samples – this includes formalin-fixed and fresh frozen tissue sections.
- Expert interpretation – a consultant pathologist assesses the immunoreactivity and tissue specificity of antibodies.
- High-throughput and cost-effective screening process for the desired clones.
Immunohistochemistry validation workflow